Lentivirus Options for SARS-CoV-2 Research
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV2) or COVID-19 is a novel respiratory coronavirus that was first isolated in December 2019 from three people connected to a cluster of pneumonia cases in Wuhan City, China. As of June 26 2020, some 6 months since the first identification, SARS CoV2 has now spread globally to nearly every country, infecting more than 9.7 million people and leading to more than 493,000 deaths. The R0 (nought/zero) of SARS CoV2, which measures its contagiousness, is 2-2.5, which means that one infected individual can spread the virus to 2 to 3 individuals. This R0 is similar to that of the 1918 influenza virus that killed over 50 million people worldwide, which makes SARS CoV2 a highly contagious virus.

LENTIVIRUS VECTOR WITH SARS-COV-2-S GENE
Tailored Genes has also developed lentivirus vector with the SARS CoV2 spike gene. Sequential transduction of target cells with different MOIs of LVV CoV2-S results in sequential increase in expression of SARS CoV2-S spike protein. The LVV CoV2-S is of high purity and of high titer (10e8 IU/mL). This vector will allow studies to explore CoV2 spike protein expression in target cells.
The LVV eGFP (CoV2-S) pseudovirus and LVV CoV2-S are available upon request.

A SAFE SARS COV2 VECTOR TO STUDY VIRAL ENTRY AND FOR TESTING ANTIVIRALS
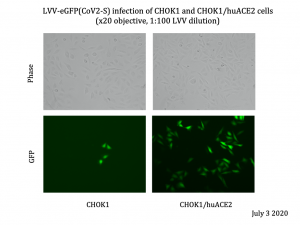
Tailored Genes has developed a SARS CoV2 vector (CoV2 vector) with an eGFP marker that can be safely used in a BSL2 facility. Their vector is a hybrid between a lentivirus vector and SARS COV2 spike protein. Their CoV2 vector also carries the eGFP gene as a marker to visualize infected cells and uses human ACE2 protein as a receptor for entry (see figure at left). Their vector is of high purity, thus increasing transduction efficiency of target cells. Because the SARS CoV2 spike protein is critical in the initial binding and infection of cells, our safe CoV2 vector will allow Institutions to study SARS CoV2 entry and allow testing of antivirals that block entry of SARS CoV2.
The publication of the genetic sequence of the SARS CoV2 in January 2020 has greatly helped the research community in understanding this pathogenic coronavirus, and has sped up progress in development of vaccines, drugs and antibodies against SARS CoV2. Developing new drugs and antibodies, collectively known as antivirals, will require testing against SARS CoV2. However, SARS CoV2 is highly contagious and use of this coronavirus requires a minimum biosafety level (BSL) 3 facility (BSL 1 being minimum containment and BSL 4 being the maximum containment). Unfortunately, BSL3 facilities are very limited and this can severely limit testing of antivirals against SARS CoV2
SARS COV2 IN RESEARCH, AND IN VACCINE AND ANTIVIRAL DEVELOPMENT

Please contact us to learn more!
Contact UsSAFETY
Tailored Genes provide 3rd Generation vectors consistent with guidelines provided by the Public Health Agency of Canada. This means they meet the highest safety standards on the market and guaranteeing transduction of targeted cells with minimum toxicity. Our 3rd Generation vectors are produced by transfection of HEK293T cells with 4 endotoxin-free expression plasmids.